|
Because of the quantized orbits, such “quantum jumps” will produce discrete spectra, in agreement with observations. The orbiting electron in Bohr’s model is assumed not to emit any electromagnetic radiation while moving about the nucleus in its stationary orbits, but the atom can emit or absorb electromagnetic radiation when the electron changes from one orbit to another. h E ( 1 n l o w 2 1 n h i g h 2) 13.6 eV. Bohr explained the hydrogen spectrum in terms of electrons absorbing and emitting photons to change energy levels, where the photon energy is. The Bohr model retains the classical mechanics view of circular orbits confined to planes having constant energy and angular momentum, but restricts these to quantized values dependent on a single quantum number, n. Bohrs model calculated the following energies for an electron in the shell, n. If classical electromagnetic theory is applied, then the Rutherford atom would emit electromagnetic radiation of continually increasing frequency (contrary to the observed discrete spectra), thereby losing energy until the atom collapsed in an absurdly short time (contrary to the observed long-term stability of atoms). This quiz and worksheet will allow you to test your skills in the following areas: Reading comprehension - ensure that you draw and retain the most important information on the Bohr model and the. Solubility Graph Worksheet Answers - Explore professionally designed templates to get your wheels spinning or create your worksheet from scratch. If the requirements of classical electromagnetic theory that electrons in such orbits would emit electromagnetic radiation are ignored, such atoms would be stable, having constant energy and angular momentum, but would not emit any visible light (contrary to observation). Bohr Atomic Models Worksheet : 19 Best Images Of Bohr Diagram Worksheet / Worksheeto Inside Bohr Atomic Models Worksheet. 
According to classical mechanics, the Rutherford model predicts a miniature “solar system” with electrons moving about the nucleus in circular or elliptical orbits that are confined to planes. Students who can answer open-ended questions about the lab truly understand. Students will have to follow the directions on the task cards and using the small squares that represent the subatomic particles (protons, neutrons, and electrons) create certain atoms. 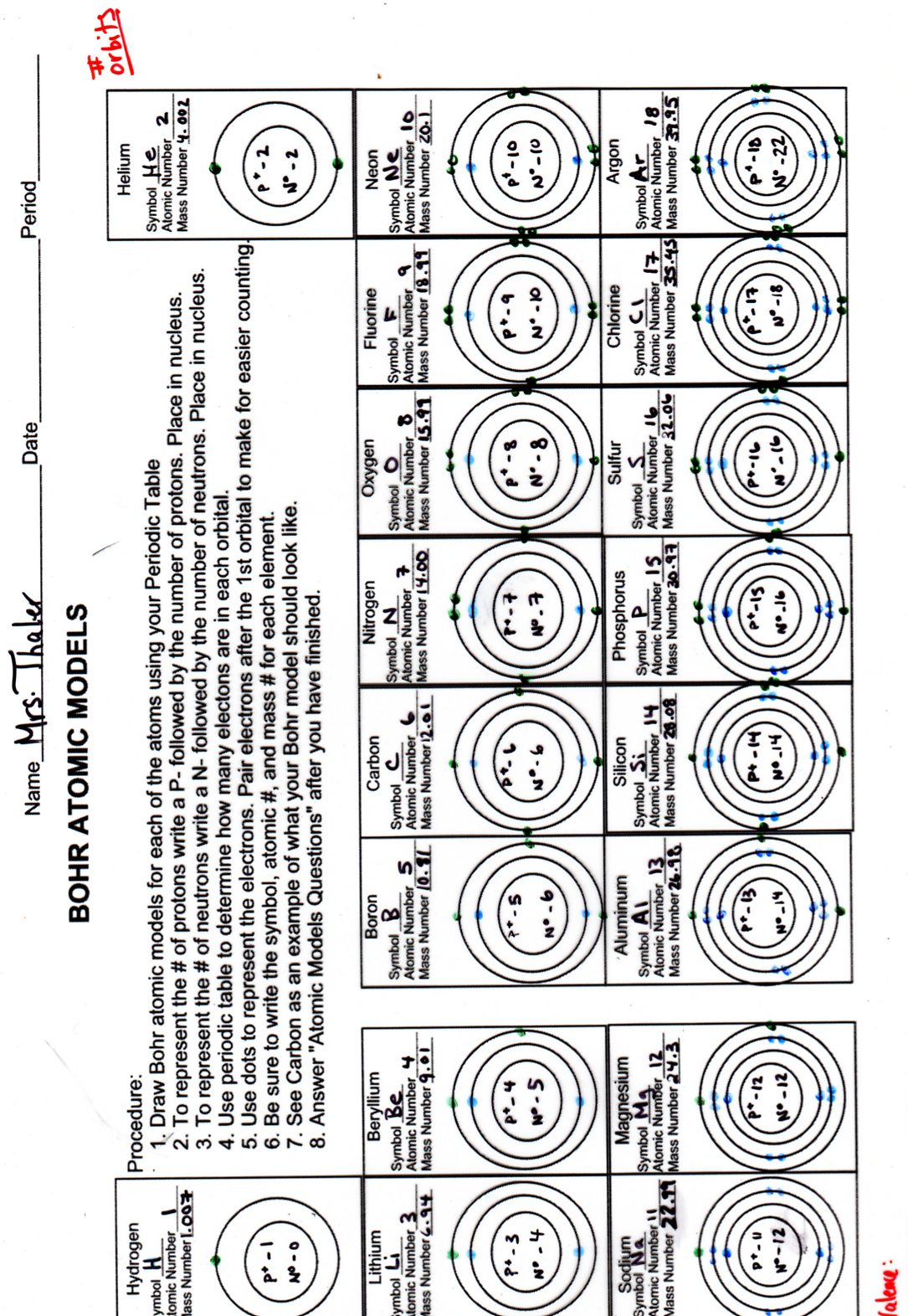 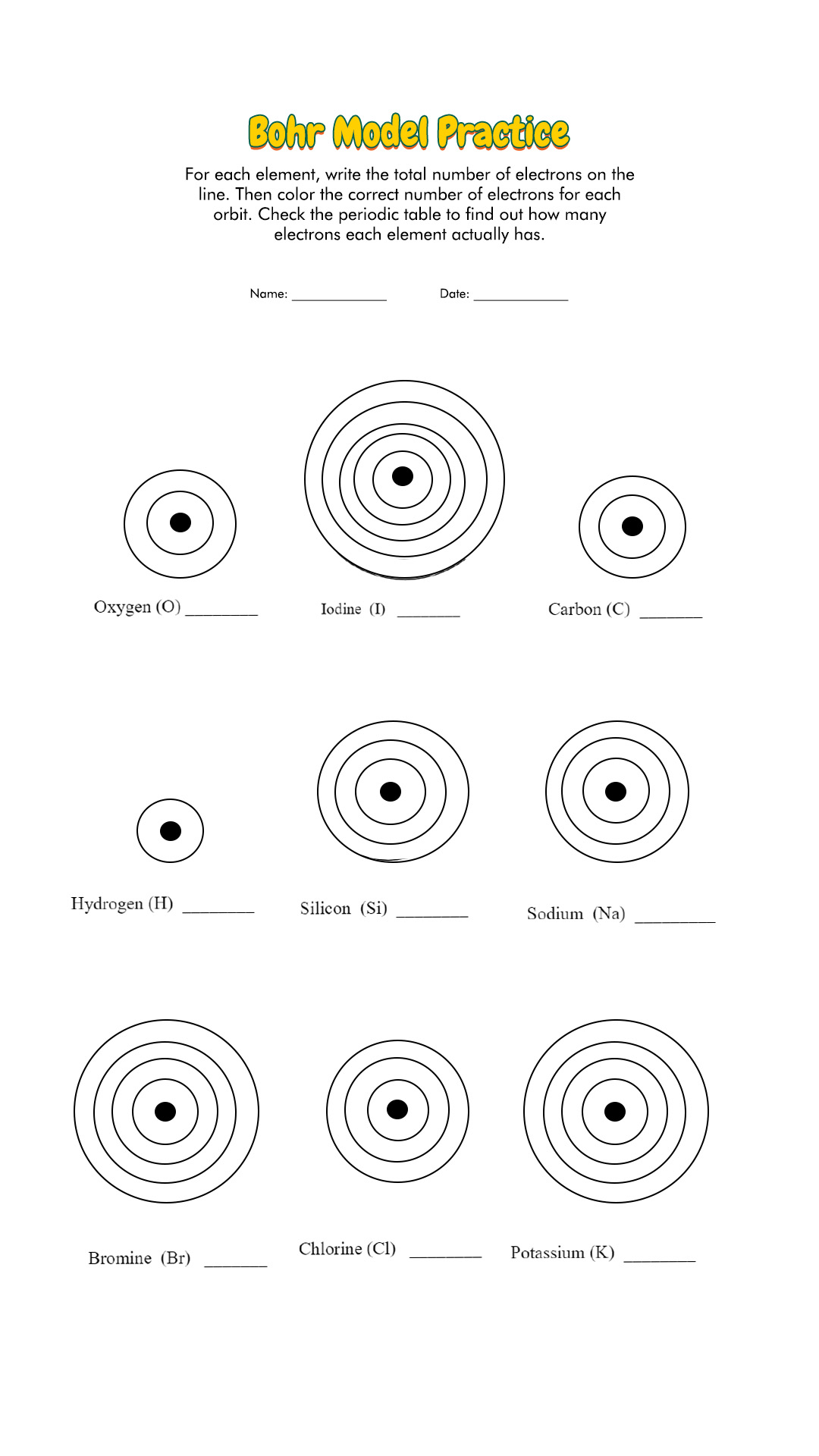
How are the Bohr model and the Rutherford model of the atom similar? How are they different?īoth involve a relatively heavy nucleus with electrons moving around it, although strictly speaking, the Bohr model works only for one-electron atoms or ions. Students will be working in pairs to create atom models using a Bohr template.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
